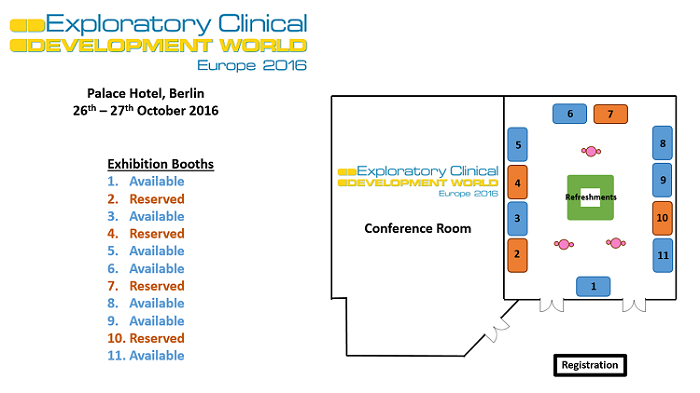
Floor plan | Book your sponsor stand now.
Exploratory Clinical Development World Europe will provide a comprehensive review of scientific and regulatory challenges in developing viable products in early stage clinical development. Our attendees are looking to alleviate the bottlenecks in early clinical development and need to find ways to avoid the pitfalls and considerable costs of taking ineffective agents into full-scale clinical development. If you are in the business of providing world-class solutions you need to be here.
Reserve your place on the sponsors' floor today and be at the forefront to showcase your innovations to pharma, biotech and healthcare professionals in PK/PD, Phase I/IIaTrials, Clinical Pharmacology, Clinical Research, Translational Medicine and more.
Health Network Communications has an excellent track record of bringing the right people and organisations together in order initiate new and progress existing relationships. We create an informative and engaging environment where the industry and the supplier community can meet and network.
This is a great opportunity to increase brand awareness and positioning, arrange meetings with top prospects and educate the Exploratory Clinical Development community on your products and services.
Your customers are here to:
-
Learn new approaches in de-risking early candidate selection
-
Improve success rates through the stimulation of open innovation and collaborations
-
Develop successful early phase clinical design strategies
-
Maximise opportunities in biomarker lead clinical development
-
Understand the role of translational medicine
-
Learn how to make accurate risk assessments in first in human trials
Who should sponsor?
-
Full Service CRO
-
Niche / Specialist CRO
-
Central Labs
-
Laboratory Services
-
Technical Services
-
Biomarker Solutioins
-
Clinical Trial Design
"Excellent Conference, high quality & authoritative presentations, tremendous depth of real-world experience ” |Medical Director, BiogenIdec
